Pharmaceuticals
Celebrating the invention of the GC-MS
10th July 2019
By Mark Jones, Executive External Strategy and Communications Fellow at Dow Chemical
In June 2019, The American Chemical Society (ACS) designated Midland, Michigan, a National Historic Chemical Landmark for the firs
In June 2019, The American Chemical Society (ACS) designated Midland, Michigan, a National Historic Chemical Landmark for the first demonstration of gas chromatography-mass spectrometry. Mark Jones, Executive External Strategy and Communications Fellow at Dow Chemical, explains why this technology is now one of the most important in the analytical chemist’s toolbox.
I like Reese’s Peanut Butter cups. I’m not much of a chocolate fan. I can take or leave peanut butter, but I find the combination delicious. Reese’s Peanut Butter cups are two great tastes that taste better together. GC-MS, gas chromatography-mass spectrometry, is the Reese’s of analytical instrumentation, mixing two technologies in a combination that is better, more powerful, than either alone. On June 8th, The American Chemical Society (ACS) designated a National Historic Chemical Landmark for the first demonstration of the powerful combination. Dow researchers Fred McLafferty and Roland Gohlke demonstrated the first pairing of gas chromatography with mass spectral detection1 in the winter of 1955 in Midland, Michigan. GC-MS is now one of the most widely deployed, most powerful technologies2 in the analytical chemist’s toolbox.
In GC-MS, chromatography separates a mixture into individual components, with each chromatographic peak being analysed by a mass spectrometer. Components are identified unambiguously when both characteristic retention times and mass spectral fingerprints match reference materials. When analysing unknown materials, the mass spectrum provides tentative identifications, dramatically reducing the number of possible matches. Because of its power and sensitivity, the technique is widely used in medicine, forensic analysis, environmental testing, drug testing and more.
GC takes advantage of the differing affinities different vapours have for surfaces. A mixture is first vapourized by rapid heating is a flow of gas. The vapour and the carrier gas are pushed into a tube that in the 1950s was packed with small particles. Properties of the particles, like porosity or nature of a coating, slow some compounds more than others. Small, light compounds get through the column faster than big, heavy ones. A detector at the end of the column produces a signal as each compound elutes or exits. Plotting the signal gives a peak for each component in the mix. Run in the same way, the pattern of peaks is reproducible. Calibrating with known compounds gives the ability to identify components in a mixture based on retention time. Powerful as it is, chromatographic retention time alone is insufficient to unambiguously identify a component in a mixture. There is always a chance that two chemicals will have the same retention time.
Roland Gohlke built the GCs used in the first GC-MS and packed his own columns. GC was still very much in its infancy in the late 1950s.
Mass spectrometry was also in its infancy in the late 1950s.3 All mass spectrometers work under vacuum. Ions are manipulated and accelerated with electric fields, ultimately hitting a detector. In 1955, mass spectrometers were sold by a few manufacturers. These instruments could identify and analyse pure materials pretty well. For every chemical, a “mass spectrum” related to the structure of the original, intact molecule is produced. Any compound will break into the same ions under the same conditions, creating a unique mass spectrum, a fingerprint that can be used to identify the material. 70 V became the standard electron energy and libraries of mass spectral fingerprints soon became available to aid in the identification of unknown materials. When two or more materials are present, the mass spectrum is a combination of the spectra of each of the components. Rather than a clear fingerprint, the result is a mess that can’t always be used to identify and quantify the components. MS is great for pure materials, but not so great for mixtures.
Magnetic sector instruments of the mid-1950s accelerated ions produced by shooting electrons through a vapour. Substances were leaked into an ion source, accelerated and directed through a magnetic field. Ions are deflected, their paths bent like light through a prism, separating them by charge. These magnetic sector instruments were cumbersome and slow. Detroit-based Bendix Corporation introduced commercial time-of-flight instruments in the mid-1950s. These pulsed an ion source and accelerated the resulting ions through a long drift tube. The velocity of any ion, and, therefore, the time it took to traverse the drift tube, are dependent on the mass-to-charge ratio. Amplifying the current resulting from the ions hitting a detector at the end of the drift tube yields a full mass spectrum for each pulse of the ion source. Mass spectra are rapidly captured on an oscilloscope, much more rapidly than a magnet could be scanned.
Fred McLafferty heard about the Bendix time-of-flight mass spectrometer and knew that its speed made coupling with a GC practical. He reasoned that adding the power of mass spectrometry to identify pure components with the power of chromatography to separate a mixture into pure components would be more than additive. Matching the retention time and mass spectra of a compound would seal the identification in a way chromatographic retention time alone could never do. Using chromatography to separate a mixture into pure components allows identification and quantitation impossible by taking a mass spectrum of a mixture.
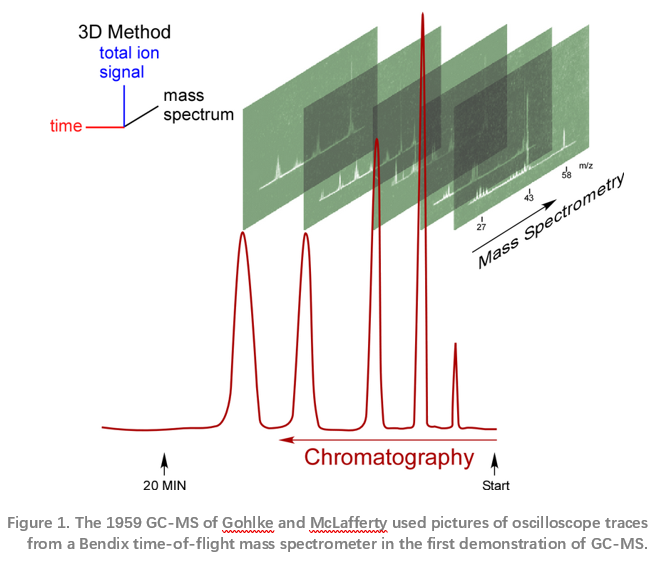
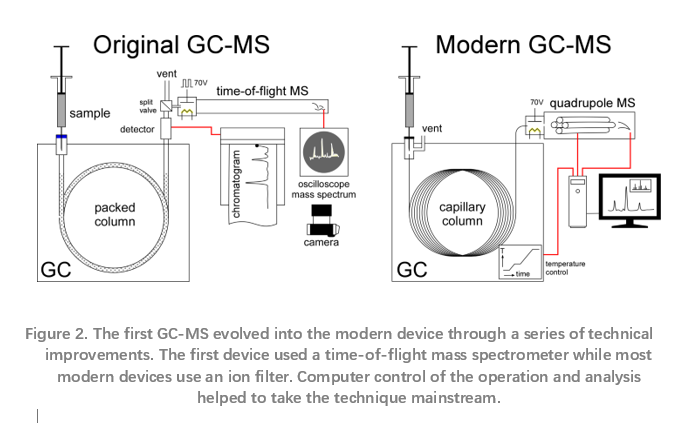
McLafferty and Gohlke faced an immediate problem – pressure. GCs operate at pressure and mass specs operate under vacuum. Their solution was to use a leak valve they made, putting only part of the GC flow into the ion source. A thermal conductivity detector recorded the chromatogram while spectra showed up only on an oscilloscope. A camera, manually triggered when a chart recorder showed a chromatographic peak was eluting, captured the mass spectra for analysis. The first paper McLafferty and Gohlke published shows photographs of the oscilloscope screen displaying the mass spectra.4 This gave way to an ultrafast chart recorder, the first of many modifications in the evolution of GC-MS to the devices of today.
Mass spectrometers work on several different principles. The Bendix MS used by Gohlke and McLafferty was a time-of-flight (TOF) instrument where the time it took ions to traverse a long tube produced a spectrum. Development of small, fast mass filters that were far superior to the TOF or magnetic sector instruments enabled the technique to really go mainstream. The introduction of the quadrupole mass spectrometer made possible smaller, less expensive GC-MS.
Computer control and spectral matching also were important to the development of GC-MS. Standardization of ionization energy meant that all MS units produce the same fingerprint for the same compound. Libraries of mass spectra were digitized so that computer matching was possible.5 GC-MS systems could now automatically identify chromatographic peaks.
Turbomolecular pumps replaced cumbersome and messy diffusion pumps. Production of oil-free vacuum systems reduced contamination and maintenance, dramatically improving operability of systems.
The widespread adoption of wall-coated, open tubular (WCOT) or capillary columns, replacing packed columns, is another important advance. These columns work well with very small amounts of material, well-suited for the great sensitivity of MS. They also provide very narrow, sharp chromatographic peaks, improving MS detection. The resolution robbing leak valve used by Gohlke and McLafferty is gone. The full column effluent enters the ion source.
The invention of the GC-MS spawned a number of other hyphenated techniques. Coupling liquid chromatography with MS gives LC-MS. GC with infrared spectroscopy gives GC-IR. The list goes on, all started with the first coupled technique, GC-MS.
Advances in both chromatography and mass filters continue with no sign of stopping. GC-MS is now the go-to technology in modern chemistry labs. Applications include development of new pharmaceuticals and analysis of their purity, detection of chemical warfare agents and explosives, screening of athletes’ urine for banned performance-enhancing substances, and checking food quality and safety. The technology has come a long way from the experiments of Gohlke and McLafferty.
Fred McLafferty provided insights in a video interview conducted by Héctor D. Abruña, Professor of Chemistry at Cornell, to open the dedication ceremony. Ron Hites, Distinguished Professor at the Indiana University, and Gustavo Serrano, Global Product Manager for Gas Chromatography Columns at Agilent Technologies spoke on the history and merits of GC-MS. They offered differing views reminiscent of the Reese’s debate. The GC is merely an adjective, just a sample introduction technique for MS in Hites’ view.6 Serrano offered that improvements in GC technology are largely responsible for the wide acceptance and adaptability of the combined technique. It was analogous to the Reese’s issue. It doesn’t matter whether it is viewed as a GC inlet to a MS or MS as a GC detector, the two techniques are simply better together. The pioneering work of Gohlke and McLafferty demonstrated that it was possible to couple GC with MS to create an exceedingly powerful tool. While they clearly saw the power in the combination, McLafferty and Gohlke never could have grasped how powerful, how ubiquitous or how important the technique would become.
References
1. Gohlke RS, McLafferty FW. J Am Soc Mass Spectrometry 1993;4:367–71.
2. Hites RA. Anal Chem 2016;88:6955–61.
3.Griffiths J. Anal Chem 2008;80:5678–83.
4. Gohlke RS. Anal Chem 1959;31:535–41.
5. McLafferty FW, Venkataraghavan R. J Chromatogr Sci 1979;17:24–9.
6. Budde WL. Environ Sci Technol 2015;49:13741–2.
Author
Mark Jones, Executive External Strategy and Communications Fellow at Dow Chemical, Midland, Michigan, USA


